Saif
Senior Member
- Jan 24, 2024
- 19,202
- 8,863
- Nation

- Residence

- Axis Group

Renata begins exporting two new medicines to the UK
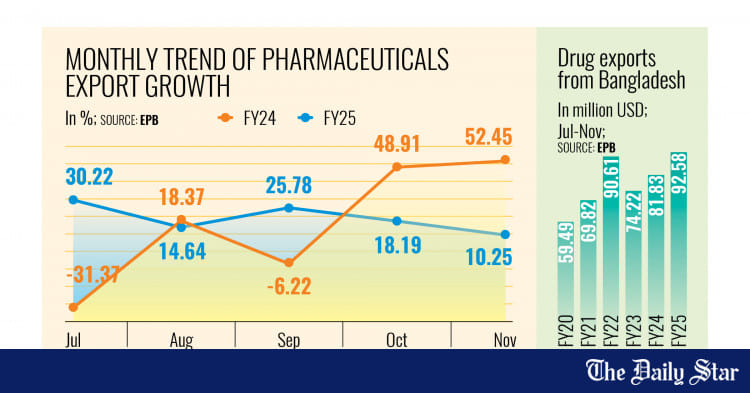
Renata PLC, a leading pharmaceutical company in Bangladesh, has started exporting two new products to the UK, thereby expanding its product portfolio in the country, according to a company disclosure.
Renata begins exporting two new medicines to the UK
Renata PLC, a leading pharmaceutical company in Bangladesh, has started exporting two new products to the UK, thereby expanding its product portfolio in the country, according to a company disclosure.
Posted on the Dhaka Stock Exchange (DSE) website, the disclosure informed that Renata shipped its first consignment of Sertraline 100 milligram (mg) and Propylthiouracil 50mg tablets to the UK yesterday.
The listed drug maker also said the two products manufactured at its facility in Gazipur will be marketed in the UK under its own brand.
Sertraline is the most commonly prescribed antidepressant in the UK market, with more than 23 million prescriptions dispensed in 2023.
It is used for certain conditions, including depression, anxiety, obsessive-compulsive disorder and post-traumatic stress disorder.
The second product, Propylthiouracil, is a key treatment for physical ailments such as hyperthyroidism, particularly in patients with Graves' disease.
The disclosure also said the addition of this product reinforces Renata's commitment to providing essential thyroid management solutions as a part of the company's chronic care portfolio in the UK.
Sertraline and Propylthiouracil are marketed in Bangladesh under the brand names Seronex and PTU respectively.
Renata PLC, a leading pharmaceutical company in Bangladesh, has started exporting two new products to the UK, thereby expanding its product portfolio in the country, according to a company disclosure.
Posted on the Dhaka Stock Exchange (DSE) website, the disclosure informed that Renata shipped its first consignment of Sertraline 100 milligram (mg) and Propylthiouracil 50mg tablets to the UK yesterday.
The listed drug maker also said the two products manufactured at its facility in Gazipur will be marketed in the UK under its own brand.
Sertraline is the most commonly prescribed antidepressant in the UK market, with more than 23 million prescriptions dispensed in 2023.
It is used for certain conditions, including depression, anxiety, obsessive-compulsive disorder and post-traumatic stress disorder.
The second product, Propylthiouracil, is a key treatment for physical ailments such as hyperthyroidism, particularly in patients with Graves' disease.
The disclosure also said the addition of this product reinforces Renata's commitment to providing essential thyroid management solutions as a part of the company's chronic care portfolio in the UK.
Sertraline and Propylthiouracil are marketed in Bangladesh under the brand names Seronex and PTU respectively.